- Home
- Services
- About
- News
- Contact
- Naruto forehead protector style
- Not seeing emulator in rslinx classic
- Easyworship 6 change browser
- Download sketchup version 8 house builder
- Dev dd season 2 watch online free
- Astute graphics plugin review
- Corel draw x6 vs x7
- Words to hotel california
- Whisper app chat
- Avg for windows server 2008 r2 64 bit
- Burnet cad
- Digimon world re digitize english psp iso
- Adobe premiere pro cc
- Sailor moon episodes 13
- The ground ola gjeilo
- Realflow crm
- Hydrophobic amino acids in alpha helix
- Mungda meaning hindi
- Where do i watch twilight online for free
- Mature komik sub indo
- Vmix 14 manual registration
- Full movie final destination 1
- Tamil vijay tv shows
- Proshow gold 9
- How to make paragraph spacing in word
- Resident evil 5 ps3 save file
- Creamware prodyssey
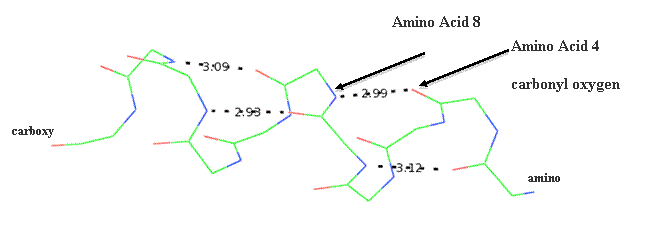
which type of interaction is not associated with tertiary structure? in which of the following proteins is the alpha-felix the predominant structure?. The amyloid protein deposition associated with Alzheimer’s disease is composed of Alpha helix Beta pleated sheets Beta bends Tertiary structure 7. the most important force is hydrophobic interaction (or hydrophobic bonds).
in which of the following proteins is the alpha-felix the predominant structure? A single polypeptide chain may contain multiple alpha-helix and beta-pleated sheet regions. Also, the R-group of each amino acid may be either hydrophobic or hydrophilic. Alpha-synuclein is a member of the synuclein family, which also includes beta- and gamma-synuclein.
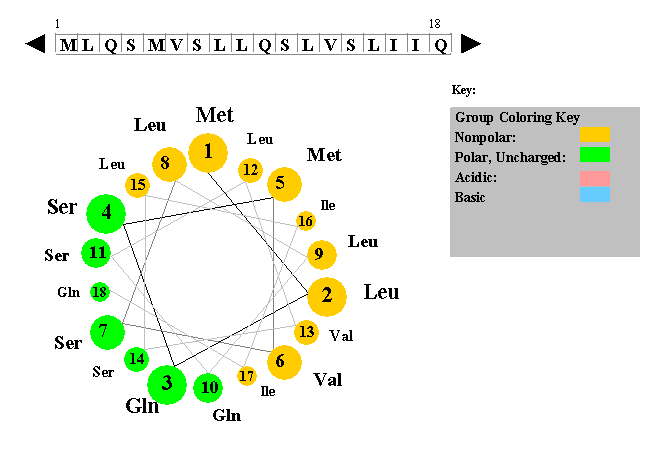
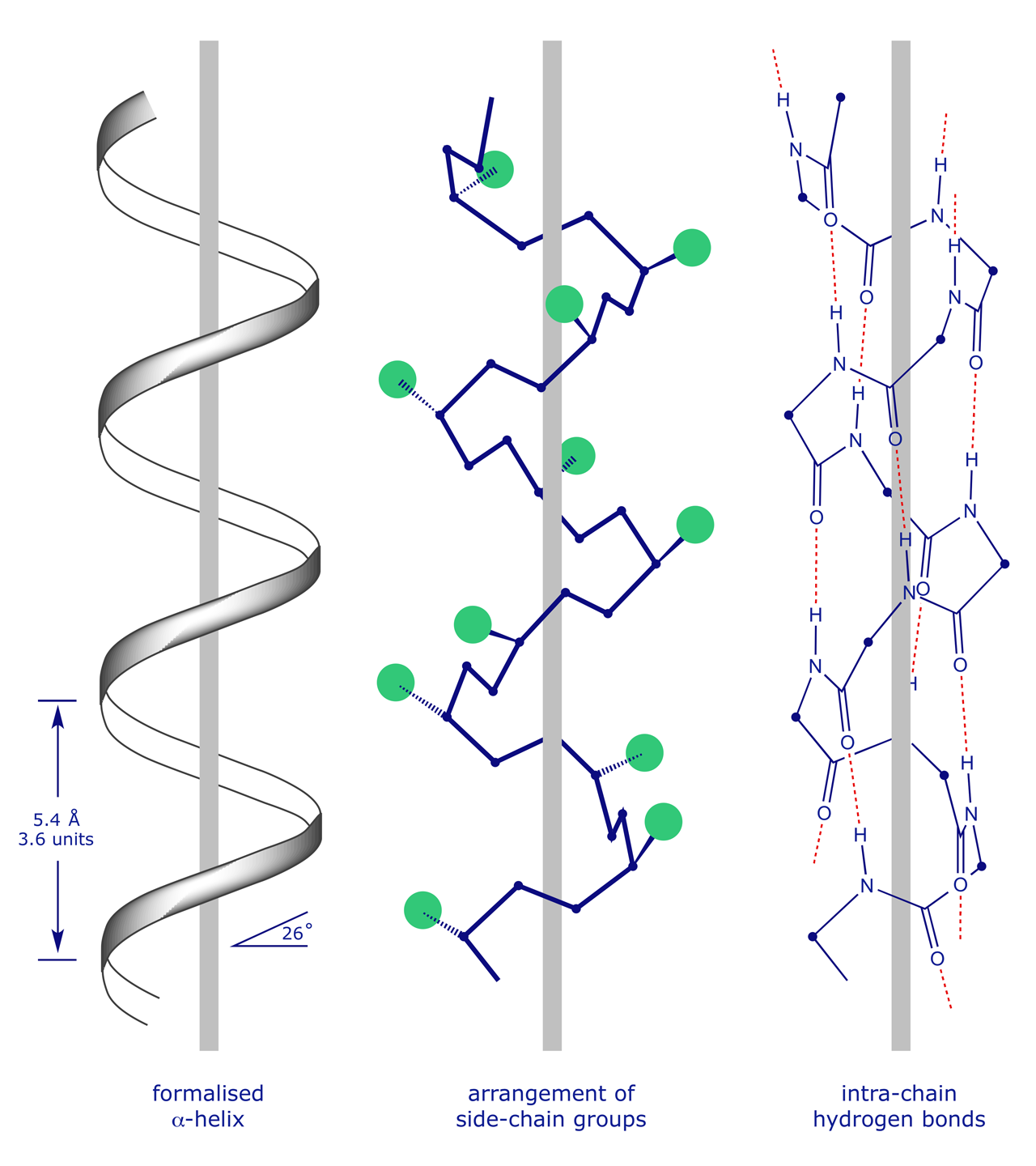
Since the fatty acid “tails” of phospholipids are nonpolar and hydrophobic and the glycerol and phosphate “heads” are polar and hydrophilic, phospholipids are often found oriented in sandwichlike formations with the hydrophobic heads oriented toward the outside. The hydrophobic side chains will tend to get away from water environment while hydrophilic side chains are attracted towards it. For instance, if a stretch of about 20 amino acids shows positive for hydrophobicity, these amino acids may be part of alpha-helix spanning across a lipid bilayer, which is composed of hydrophobic fatty acids. The exact angle of helix packing depends on the sequence of the protein, because packing is mediated by the sterics and hydrophobic interactions of the amino acid side chains near the helix interfaces. Defects in SNCA have been implicated in the pathogenesis of Parkinson disease. The R groups (the side chains) of the polypeptide protrude out from the α-helix chain and are not involved in the H bonds that maintain the α-helix structure. One type is the alpha (α) helix structure. This makes an alpha-helix long enough to span a membrane. Nonpolar molecules are repelled by water and do not dissolve in water are hydrophobic. a) nonpolar covalent bonds b) hydrophobic interactions c) peptide bonds d) hydrogen bonds e) ionic bonds Some proteins are made of subunits in which protein molecules bond together to form a larger unit.

SNCA may serve to integrate presynaptic signaling and membrane trafficking. The alpha helix has a right handed helix conformation. R group can be hydrophobic or hydrophilic. There are almost 20 amino acids are found in human body that varies in their R groups. alpha-helix, beta-sheet and beta-turn) Avoidance of specific sequence motifs (ex. Every helical turn in an alpha helix has 3.6 amino acid residues. Hydrophobic interactions greatly contribute to the folding and shaping of a protein.The "R" group of the amino acid is either hydrophobic or hydrophilic. Enzymes are more often globular with hydrophilic amino acids on the outside and hydrophobic amino acids folded in towards the middle. In arrays of membrane-spanning helices, helices in the interior of the array could be shorter. This structure resembles a coiled spring and is secured by hydrogen bonding in the polypeptide chain. Which type of interaction stabilizes the alpha helix and the beta pleated sheet structures of proteins? Tertiary structure is the three-dimensional structure of a protein. Tertiary structure is the next level of complexity in protein folding. Which of the following is the most common and stable conformation for a polypeptide chain Alpha helix Beta pleated sheets Anti parallel beta pleated sheet Tertiary structure 6. leaving hydrophilic amino acids on the outside to interact with surrounding water molecules. Synucleins are abundantly expressed in the brain and alpha- and beta-synuclein inhibit phospholipase D2 selectively.
- Home
- Services
- About
- News
- Contact
- Naruto forehead protector style
- Not seeing emulator in rslinx classic
- Easyworship 6 change browser
- Download sketchup version 8 house builder
- Dev dd season 2 watch online free
- Astute graphics plugin review
- Corel draw x6 vs x7
- Words to hotel california
- Whisper app chat
- Avg for windows server 2008 r2 64 bit
- Burnet cad
- Digimon world re digitize english psp iso
- Adobe premiere pro cc
- Sailor moon episodes 13
- The ground ola gjeilo
- Realflow crm
- Hydrophobic amino acids in alpha helix
- Mungda meaning hindi
- Where do i watch twilight online for free
- Mature komik sub indo
- Vmix 14 manual registration
- Full movie final destination 1
- Tamil vijay tv shows
- Proshow gold 9
- How to make paragraph spacing in word
- Resident evil 5 ps3 save file
- Creamware prodyssey
